Uranium-Thorium Dating
U-Th dating is based on the analysis of the parent (Uranium) and daughter (Thorium) isotope activity ratios. This ratio is used to approximate the disintegration of the parent and the subsequent production of the daughter over time. This is completed using mass spectrometry by measuring both the parent (234U) and daughter (230Th) products of decay in a given sample. When uranium decays, it goes through a series of decay steps (beginning in 238U) until it eventually reaches a stable isotope (ending in 206Pb, see below).
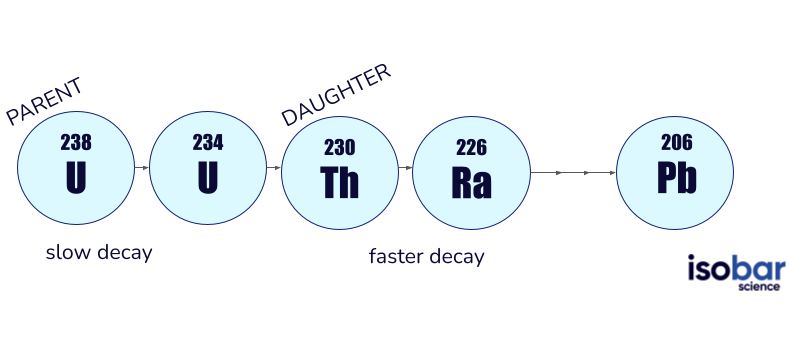
U-Th dating measures the activity ratios of parent (Uranium) and product (Thorium) isotopes, by calculating the disintegration of the parent to the daughter over time.
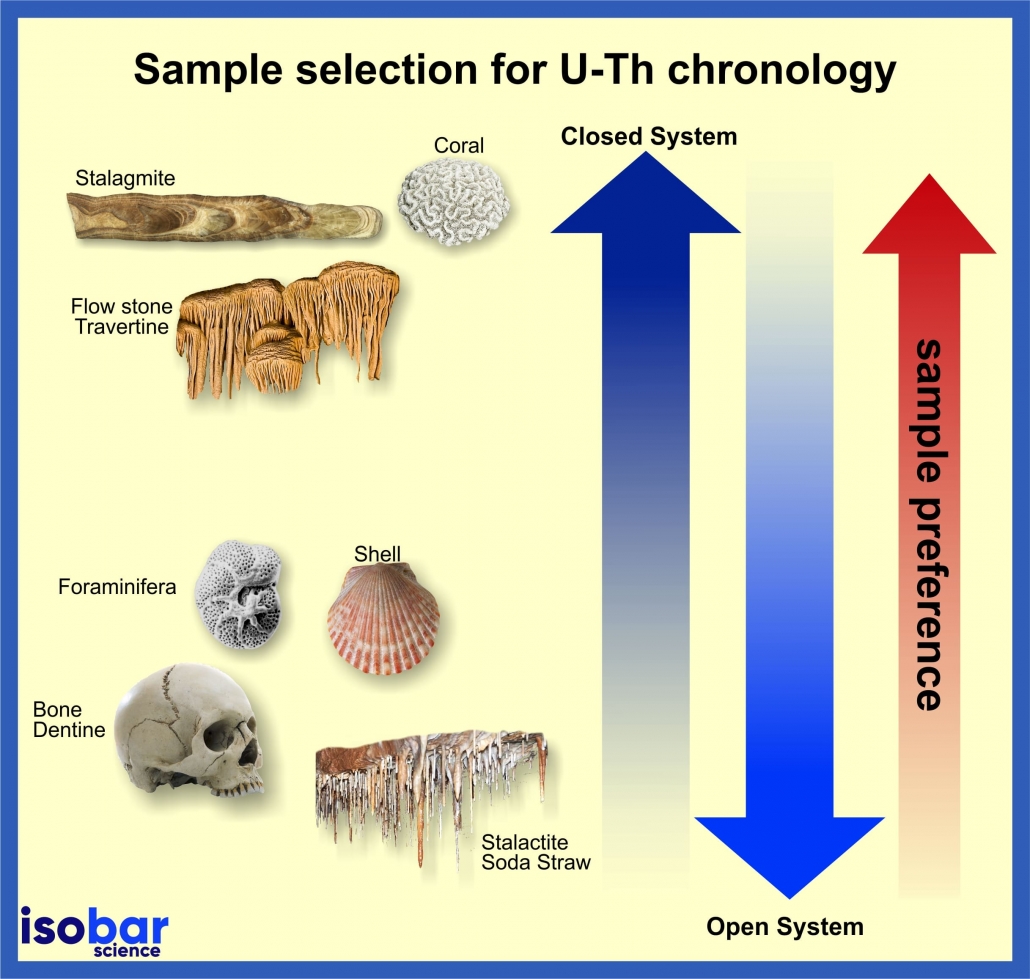
U-Th dating of carbonate samples can be conducted if the following conditions are met:
- Formed within the datable range of the U-Th systematics
(up to 500 ka), - Contain sufficient concentrations of the minor
isotopes 234U and 230Th for high-precision isotope dilution
analysis, - Incorporation of negligible or known amounts of initial
(excess) 230Th at the time of deposition, and - Remained closed with respect to U and Th isotopes since
primary deposition.
Uranium loss and inherited 230Th in open-system settings can result in erroneous conclusions. Samples with a relatively low initial U/Ca and uranium concentration tend to uptake uranium after deposition and through diagenetic processes – yielding aberrant ages.
Read more:
U-Th Dating
The U-Th method is used to date carbonate materials up to 500,000 years. Modern samples will not produce reliable U-Th dates due to their low 230Th concentrations and method limitations. Most common sample types include cave art, speleothems and flowstones, coral, and shell.
Cave Art
Uranium-Thorium analysis can provide information on the approximate date of cave art paintings by dating the thin calcite layers formed along the top of the artwork. In doing so, this analysis can provide a minimum age, also known as the terminus ante quem. Furthermore, in cases where the supporting calcite layer underlying the painting is accessible, a maximum age (terminus post quem) can also be established (Pons-Branchu et al. 2014). Results can also be verified by cross-dating the U-Th dates with 14C in addition to analyzing multiple layers along the artwork surface, due to the open system nature of carbonate crusts (Sauvet et al. 2015).

(Photo Credit: Arash Sharifi)
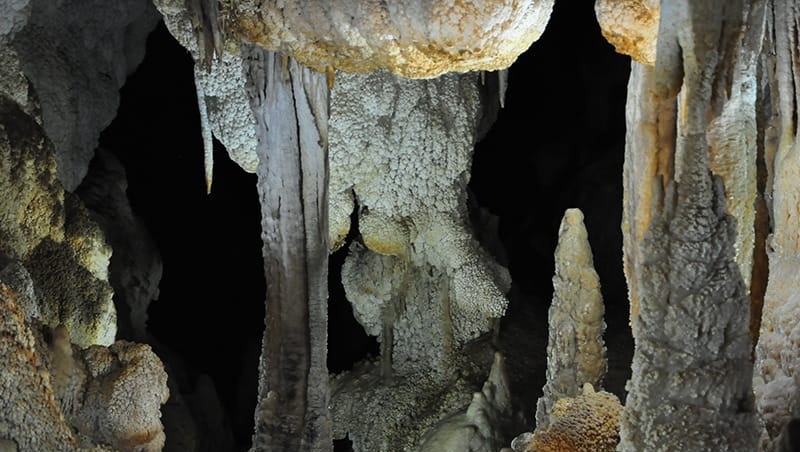
Cave Deposits: Speleothems and Flowstones
The analysis of stable isotopes in speleothems can provide information on environmental change over the course of speleothem growth. Chronologies that underlie these paleoclimate records rely on precise dating through the U-Th and/or 14C method, which provides a relationship between distance (growth) and age. Stalagmites (upward growing speleothem) are often preferred over stalactite (downward growing speleothem) due to the more regular growth plane with a more defined internal structure (Spötl & Boch, 2019). While both U-Th and 14C methods can be used to date speleothems, the reservoir effect of carbon and speleothem chemical impurities can introduce significant age uncertainties (several thousand years) in 14C dated compared to U-Th dated series (Goslar et al. 2000).
(Photo Credit: Arash Sharifi)
Corals
Corals can be dated using U-Th analysis up to 500,000 years before present. Corals have been shown to gain and lose uranium and thorium, generating an age bias. When U gain is paired with Th loss, the samples have a bias towards younger ages, whereas when U loss is paired with Th gain, the samples have a bias towards older ages (Andersen et al. 2009). Yet, systematic corrections can be applied using a screening approach (e.g. Thompson et al., 2003). The reconstruction of environmental changes through time requires a robust chronology. U-Th dating of corals provides reliable ages beyond the 14C range. These samples can be further analyzed to provide many paleoenvironmental reconstructions, including: wet/dry phases (Yehudai et al. 2017), mortality/recovery of coral reef systems (Clark et al. 2017), seawater temperature (DeCarlo et al. 2016) and pH (Stewart et al. 2016; Pauly et al. 2015).

Shells
U-Th dating has been used extensively on shell samples for decades. This method relies on the sample having incorporated uranium during formation and has limited thorium input from external sources (closed system), leaving all thorium within the structure being a product of U-Th decay. Not all depositional environments can fulfil such prerequisites, in which case shell “isochron dating” can be applied by dating multiple shells from the same time horizon with various levels of detrital Th input (Bischoff & Fitzpatrick, 1991) to obtain initial level of U-Th for dating (e.g. Placzek et al. 2006). While the open system nature of shell systems makes dating challenging, U-Th analysis offers the potential to date shells beyond the timespan of 14C dating (ca. 40,000 cal BP).

(Photo Credit: Arash Sharifi)
References
Andersen, M.B., Gallup, C.D., Scholz, D., Stirling, C.H. and Thompson, W.G., (2009). U-series dating of fossil coral reefs: consensus and controversy. Pages News, 17, pp.54-56.
Bischoff, J.L. and Fitzpatrick, J.A., (1991). U-series dating of impure carbonates: an isochron technique using total-sample dissolution. Geochimica et Cosmochimica Acta, 55(2), pp.543-554. DOI: 10.1016/0016-7037(91)90011-S
Clark, T.R., Roff, G., Zhao, J.X., Feng, Y.X., Done, T.J., McCook, L.J. and Pandolfi, J.M., (2017). U-Th dating reveals regional-scale decline of branching Acropora corals on the Great Barrier Reef over the past century. Proceedings of the National Academy of Sciences, 114(39), pp.10350-10355. DOI: 10.1073/pnas.1705351114
DeCarlo, T.M., Gaetani, G.A., Cohen, A.L., Foster, G.L., Alpert, A.E. and Stewart, J.A., (2016). Coral Sr‐U thermometry. Paleoceanography, 31(6), pp.626-638. DOI: 0.1002/2015PA002908
Goslar, T., Hercman, H. and Pazdur, A., (2000). Comparison of U-series and radiocarbon dates of speleothems. Radiocarbon, 42(3), pp.403-414. DOI: 10.1017/S0033822200030332
Pauly, M., Kamenos, N.A., Donohue, P. and LeDrew, E., (2015). Coralline algal Mg-O bond strength as a marine p CO2 proxy. Geology, 43(3), pp.267-270. DOI: 10.1130/G36386.1
Placzek, C., Patchett, P.J., Quade, J. and Wagner, J.D., (2006). Strategies for successful U‐Th dating of paleolake carbonates: An example from the Bolivian Altiplano. Geochemistry, Geophysics, Geosystems, 7(5). DOI: 10.1029/2005GC001157
Pons-Branchu, E., Bourrillon, R., Conkey, M.W., Fontugne, M., Fritz, C., Gárate, D., Quiles, A., Rivero, O., Sauvet, G., Tosello, G. and Valladas, H., (2014). Uranium-series dating of carbonate formations overlying Paleolithic art: interest and limitations. Bulletin de la Société préhistorique française, pp.211-224. DOI: 10.3406/bspf.2014.14395
Sauvet, G., Bourrillon, R., Conkey, M., Fritz, C., Gárate-Maidagan, D., Vilá, O.R., Tosello, G. and White, R., (2017). Uranium–thorium dating method and Palaeolithic rock art. Quaternary International, 432, pp.86-92. DOI: 10.1016/j.quaint.2015.03.053
Spötl, C. and Boch, R., (2019). Uranium series dating of speleothems. In Encyclopedia of caves (pp. 1096-1102). Academic Press.
Stewart, J.A., Anagnostou, E. and Foster, G.L., (2016). An improved boron isotope pH proxy calibration for the deep-sea coral Desmophyllum dianthus through sub-sampling of fibrous aragonite. Chemical Geology, 447, pp.148-160. DOI: 10.1016/j.chemgeo.2016.10.029
Thompson, W.G., Spiegelman, M.W., Goldstein, S.L. and Speed, R.C., (2003). An open-system model for U-series age determinations of fossil corals. Earth and Planetary Science Letters, 210(1-2), pp.365-381. DOI: 10.1016/S0012-821X(03)00121-3
Yehudai, M., Lazar, B., Bar, N., Kiro, Y., Agnon, A., Shaked, Y. and Stein, M., (2017). U–Th dating of calcite corals from the Gulf of Aqaba. Geochimica et Cosmochimica Acta, 198, pp.285-298. DOI: 0.1016/j.gca.2016.11.005
Coral Image: https://www.pexels.com/photo/corals-920161/
Page last updated: November 2019