Methodology: Boron Isotope Analysis
Pretreatment
Extraction chromatography using boron-specific chelating resin (Amberlite IRA-743) following Giri et al., 2019. Amberlite IRA-743 designed to remove boric acid and borate from liquid.
Additional options: boron concentration (for an additional fee in conjunction with isotope measurements) and Sample cleaning (corals, shells, carbonates)
Sample types available for boron isotope analysis: shells, corals, carbonates and water
More information on Sample Types and Selection for boron analysis
Boron Isotopes Measurement
Device: Neptune Plus multi-collector inductively coupled plasma mass spectrometer (MC-ICP-MS) with a self-aspirating nebulizer following Giri et al. (2019) and Foster et al. (2013).
Boron Isotope Analysis Results
Reported as delta (δ) values in units of parts per thousand (per mill) (‰).
Boron isotope ratios (δ11B) are reported relative to NIST SRM 951a – Boric Acid Isotopic Standard. Presented graphically on a plot which includes representative ranges of the boron isotopic composition (δ11B) of various materials.
Uncertainties for each sample measurement is presented as the 95% confidence interval (CI). Exact methods and confidence intervals will be outlined in your sample report.
Note: The lab offers boron concentration for an additional fee only in conjunction with the isotope measurements.
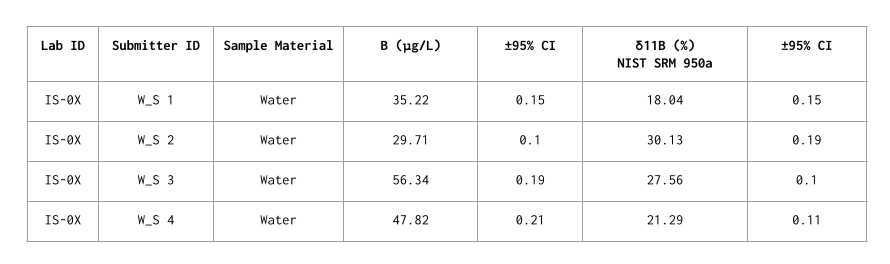
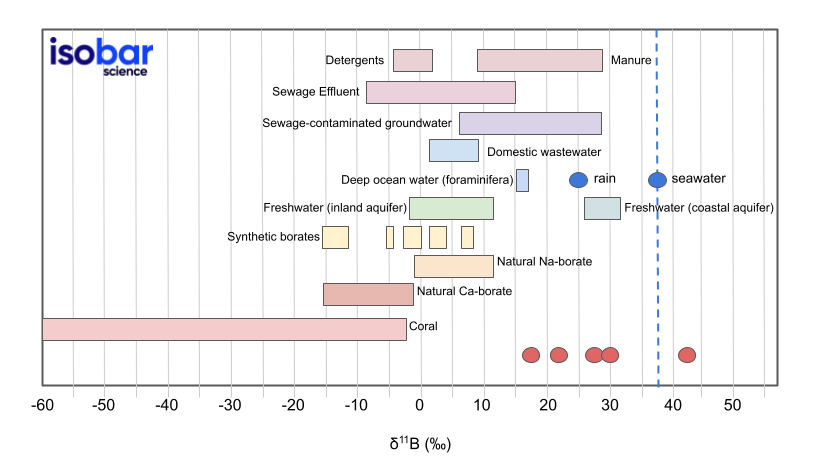
Plot of δ11B results for the tested samples (red circles) compared to a range of δ11B values from different materials in different environments.
Disclaimer: This video is hosted in a third-party site and may contain advertising.
This video excerpt is part of Beta Analytic’s webinar: Boron Isotopic Analysis
References
Foster, G. L.; Hönisch, B.; Pari, G.; Dwyer, G. S.; Rae, J.W.B.; Elliott, T.; Gaillardet, J.; Hemming, N. G.; Louvat, P.; Vengosh, A., (2013). Interlaboratory comparison of boron isotope analyses of boric acid, seawater and marine CaCO3 by MC-ICPMS and NTIMS. Chemical Geology, 358, pp. 1–14. https://www.sciencedirect.com/science/article/abs/pii/S0009254113003744?via%3Dihub
Giri, S.J., Swart, P.K. and Pourmand, A., (2019). The influence of seawater calcium ions on coral calcification mechanisms: Constraints from boron and carbon isotopes and B/Ca ratios in Pocillopora damicornis. Earth and Planetary Science Letters, 519, pp.130-140. DOI: 10.1016/j.epsl.2019.05.008
Page last updated: January 2020
