Strontium Isotope Analysis – Sample Types and Selection
The following recommendations are general guidelines. Individual sample size requirements largely vary based on the sample type. Please feel free to contact us for advice if you are concerned that your samples are too small.
Reminders:
DO NOT wrap samples in aluminum or any metal foil. Use weighing paper if you need to wrap them.
Submit samples in a ziplock bag or teflon container with PRINTED SAMPLE ID attached outside the bag.
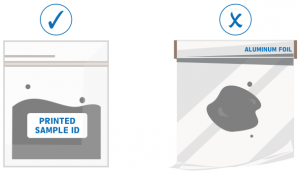
Always choose the best samples for your research objectives
Our recommended sample sizes assume the material is dry and free of adhering matrix.
Black Shale
200 mg
RECOMMENDED CONTAINERPlastic or glass vials
Put them inside a labeled Ziplock bag
Black Shale must be homogenized prior to submission.
Bone (non-ashed)
500 mg
RECOMMENDED CONTAINERZiplock bags
For the best results, make sure your samples are clean and free from soil, clay particles, calcite and other detritus.
ALSO AVAILABLE:Bone (ashed)
100-150 mg
RECOMMENDED CONTAINERVials
(preferably with screw tops)
Microcentrifuge tubes, or counting slides
For the best results, make sure your samples are clean and free from soil, clay particles, calcite and other detritus. Ash samples in a quartz crucible at 850°C for 3 hours and transfer the residues into clean small glass or plastic vials. A larger sample size is required if we need to ash the samples. Samples are digested in 8 M nitric acid (lower concentrations may be used if the sample contains clay and detritus). if the sample contains clay and detritus). A larger sample size is required if we need to ash the samples.
ALSO AVAILABLE:Bulk Carbonates
100-150 mg
RECOMMENDED CONTAINERPlastic or glass vials
Put them inside a labeled Ziplock bag
For the best results, make sure your samples are clean and free from soil, clay particles, calcite and other detritus. Samples should be homogenized prior to submission.
Coal
500 mg
RECOMMENDED CONTAINERVials, microcentrifuge tubes, counting slides
Put them inside a labeled Ziplock bag
For the best results, make sure your samples are clean and free from soil, clay particles, calcite and other detritus. Samples should be homogenized prior to submission.
Ceramics
100-150 mg
RECOMMENDED CONTAINERPlastic or glass vials
Put them inside a labeled Ziplock bag
Ceramic samples need to be homogenized prior to submittal. For effective sample dissolution, high-temperature flux fusion is the method of choice. Homogenized samples will be fused at 1075°C with high-purity lithium metaborate (LiBO2, Puratronic) and ultra-pure grade lithium bromide to ensure complete recovery of the melt. The melt is directly digested in 6 M nitric acid.
ALSO AVAILABLE:Forams (pre-extracted)
10 mg
RECOMMENDED CONTAINERVials
(preferably with screw tops)
Microcentrifuge tubes, or counting slides
For the best results, make sure your samples are clean and free from clay particles and other detritus. Samples are digested in 6 M nitric acid (lower concentrations may be used if the sample contains clay and detritus).
ALSO AVAILABLE:- None
Gypsum
150 mg
RECOMMENDED CONTAINERPlastic or glass vials
Gypsum microcrystals and gypsiferous (gypsum-rich) sediments: Gypsiferous sediments and gypsum microcrystals contain large quantities of detritic materials. Isolation of gypsum crystals from the matrix fragments must be conducted prior to sample submission. If this is not feasible by the client, Isobar Science offers isolation of gypsum crystals by means of density separation for an additional charge.
Must be received dry and homogenized or extra charge will be applied.
ALSO AVAILABLE:Igneous and Metamorphic Rocks
30-50 mg
RECOMMENDED CONTAINERPlastic or glass vials
Rock samples need to be homogenized prior to submission. For effective sample dissolution, high-temperature flux fusion is the method of choice. Homogenized samples are fused at 1075°C with high-purity lithium metaborate (LiBO2, Puratronic) and ultra-pure grade lithium bromide to ensure complete recovery of the melt. The melt is directly digested in 6 M nitric acid. Sr isotope measurements can be conducted on smaller sample sizes, please consult the lab before submitting samples.
ALSO AVAILABLE:Mineral Dust (ashed)
30-50mg
RECOMMENDED CONTAINERVials
(preferably with screw tops)
Microcentrifuge tubes, or counting slides
Aerosol samples collected on filter paper must be ashed entirely prior to submission. All organics must be removed from the sample either by ashing or with hydrogen peroxide. A larger sample size is required if we need to remove the organics or ash the samples. For effective sample dissolution, high-temperature flux fusion is the method of choice. Aerosol samples are fused at 1075°C with high-purity lithium metaborate (LiBO2, Puratronic) and ultra-pure grade lithium bromide to ensure complete recovery of the melt. The melt is directly digested in 6 M nitric acid. Sr isotope measurements can be conducted on smaller sample sizes, please consult the lab before submitting samples.
ALSO AVAILABLE:Peat
300 mg
RECOMMENDED CONTAINERPlastic or glass vials
Put them inside a labeled Ziplock bag
All organics must be removed prior to submission.
Plant and Woods
1-2 g (1000-2000 mg)
RECOMMENDED CONTAINERZiplock bags
For the best results, make sure your samples are clean and free from soil, clay particles, calcite and other detritus.
ALSO AVAILABLE:- None at this time
Sediments (detrital, carbonate, or bulk)
50 mg
RECOMMENDED CONTAINERPlastic or glass vials
Put them inside a labeled Ziplock bag
All organics must be removed from the sample either by ashing or with hydrogen peroxide. All carbonate fractions must be removed if you wish to measure Sr isotopes only on siliciclastic fraction. A larger sample size is required if we need to remove the carbonates and organics from the samples. For effective sample dissolution, high-temperature flux fusion is the method of choice. Sediment samples will be fused at 1075°C with high-purity lithium metaborate (LiBO2, Puratronic) and ultra-pure grade lithium bromide to ensure complete recovery of the melt. The melt is directly digested in 6 M nitric acid. Sr isotope measurements can be conducted on smaller sample sizes, please consult the lab before submitting samples.
ALSO AVAILABLE:Shells, Corals &
Bulk Carbonates
100-150 mg
RECOMMENDED CONTAINERZiplock bags
For the best results, make sure your samples are clean and free from clay particles and other detritus. Homogenize your samples, preferably using an agate mortar and pestle, and transfer into plastic or glass vials. Homogenized samples are digested in 6 M nitric acid (lower concentrations may be used if the sample contains clay and detritus).
ALSO AVAILABLE:Soil
150mg (ashed), 500mg (non-ashed)
RECOMMENDED CONTAINERPlastic or glass vials
Put them inside a labeled Ziplock bag
All organics must be removed and samples must be homogenized prior to submission.
Stalagmite and Flowstone
100 mg
RECOMMENDED CONTAINERGlass or plastic vials
Homogenize your samples, preferably using an agate mortar and pestle, and transfer into plastic or glass vials. Homogenized samples are digested in 6 M nitric acid.
ALSO AVAILABLE:Silk
100-150 mg
RECOMMENDED CONTAINERZiplock bags
For the best results, make sure your samples are clean and free from soil, clay particles, calcite, and other detritus.
ALSO AVAILABLE:- None at this time
Tooth Enamel/Dentine
100 mg
RECOMMENDED CONTAINERVials
(preferably with screw tops)
Microcentrifuge tubes, or counting slides
To remove non-biogenic Sr from enamel or bone, samples will be sequentially treated by 0.1 N acetic acid. Please contact the lab if you want to follow different protocols or you prefer to skip this stage.
ALSO AVAILABLE:Water
125 mL
RECOMMENDED CONTAINERSmall-neck plastic bottle
(HDPE, LDPE, PP)
DO NOT use glass vials
Rinse the bottle with the running sample water prior to collection. Do not add any chemicals to the water, do not acidify the water. Fill the bottle all the way to the top, you may leave a small space at the top (keep the neck of the bottle empty) to allow for any necessary expansion during shipment. Please put the bottles inside a plastic bag and seal the bag with a zip-tie or duct tape. Clearly label both the bottle and the plastic bag.
ALSO AVAILABLE:Wool
75 mg
RECOMMENDED CONTAINERZiplock bags
For the best results, make sure your samples are clean and free from soil, clay particles, calcite, and other detritus. Dye will be removed from the wool prior to digestion and extraction chromatography. Contact the lab if you are also interested in measuring strontium isotopes in dye fraction.
ALSO AVAILABLE:- Strontium Isotopes (dye fraction)
The lab also offers strontium concentration measurements for an additional fee and ONLY in conjunction with isotope analyses.
Please consult the lab before submitting samples.
Duration: 2 minutes, 40 seconds || Speaker: Arash Sharifi, PhD
This video excerpt is part of Isobar Science’s webinar: Geochemistry and Application of Strontium Isotopes.
Disclaimer: This video is hosted in a third-party site and may contain advertising.
Page last updated: November 2019
